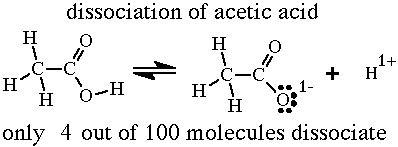
dissociation of vinegar?An everyday example of a system that is at dynamic equilibrium is the dissociation of vinegar in water, in this reaction, vinegar and water are mixed, and the two react to create hydronium ions and acetate ions. At the same time the reverse reaction is also occurring, the hydronium and acetate ions are forming acetic acid (vinegar) and water. These opposite reactions are occurring at the same rate; therefore the system is in equilibrium. This reaction is represented by the following equation:
CH3COOH(aq) + H2O(l) → CH3COO-(aq)+ H3O+(aq) |