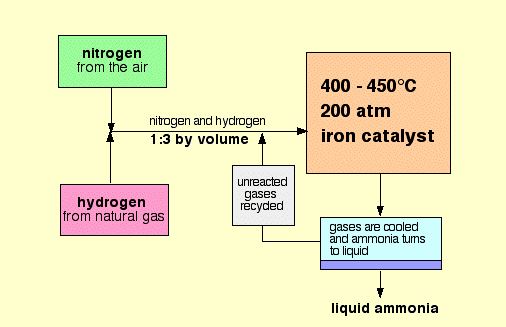
What Is The Haber Process?The Haber Process is the industrial implementation of the reaction of nitrogen and hydrogen gas. In other words, it is the main industrial process used to create ammonia. Fertilizer generated from ammonia produced by the Haber process is estimated to be responsible for sustaining one-third of the Earth's population.
Overall Process: 3 H2(g) + N2(g) → 2 NH3(g) The hydrogen used in the Haber Process comes from the reaction of methane (CH4) and steam (H2O). This reaction creates hydrogen, as well as carbon monoxide as a bi-product. This is referred to as steam-methane reforming. Getting Hydrogen Gas: CH4(g) + H2O(g) → 3 H2(g) + CO(g) The hydrogen produced in the previous reaction also reacts with the oxygen in the air to produce water, leaving behind the nitrogen in the air. The nitrogen and hydrogen gas are compressed into a reactor. This reaction occurs at a pressure of about 200atm in order to allow the reaction to favor the side of the reaction with less moles, being the product (ammonia). The reaction also occurs at a reasonably high temperature of approximately 450 degrees Celsius. This is a compromised temperature which is specifically used to ensure a good yield as well as a quick reaction time. To make the reaction reach equilibrium even quicker, an iron catalyst is present. The catalyst has no effect on the reaction itself, but does speed up the process. The overall yield of ammonia in the Haber Process is about 20%, but any reactants that are not converted into ammonia are recycled and reused. |
|