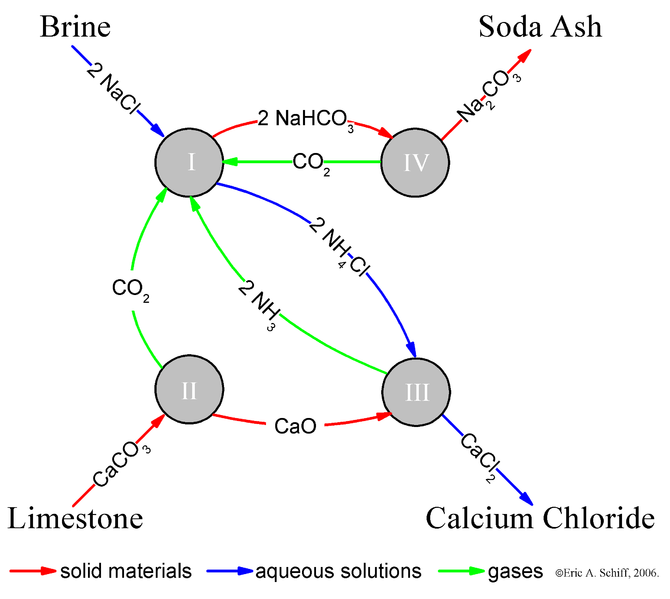
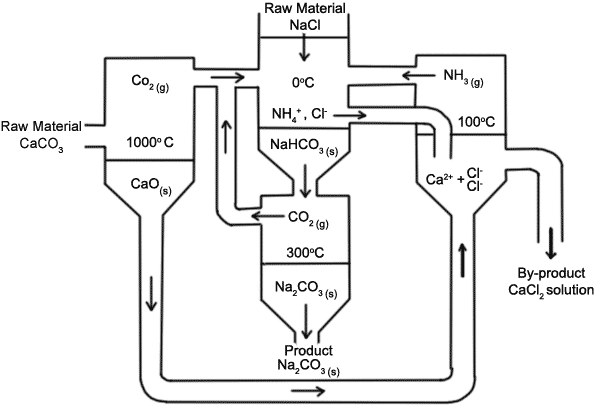
What is the solvay process?The Solvay Process is a very large process that is widely used. In fact, sodium carbonate in among the top ten inorganic chemicals in terms of quantities used.The Solvay Process produces soda ash (sodium carbonate) from brine (NaCl) and limestone (CaCO3).
Overall Reaction: 2 NaCl + CaCO3 → Na2CO3 + CaCl2 The reaction is not a simple one step process however, and must go through a variety of steps. The first step in the Solvay Process involves CO2 passing through a concentrated aqueous solution of Brine and NH3. In industrial practice, the reaction is performed by passing brine through two towers. In the first, ammonia bubbles up through the brine and is absorbed by it. In the second, CO2 bubbles up through the ammoniated brine, and sodium bicarbonate (NaHCO3) is the resulting precipitate. The ammonia buffers the solution and keeps it at a basic pH. Without the ammonia buffer, a hydrochloric acid byproduct would make the solution acidic, and not allow for the precipitate to form. Step 1: NaCl + CO2 + NH3 + H2O → NaHCO3 + NH4Cl The necessary ammonia "catalyst" for the first step is reused in a later step, and a relatively small amount of ammonia is consumed. The carbon dioxide required for the first step is produced by heating "calcination" of the limestone at 950 - 1100 degrees celsius. The calcium carbonate (CaCO3) in the limestone is partially converted to quicklime (calcium oxide (CaO) and carbon dioxide: Step 2: CaCO3 → CO2 + CaO The sodium bicarbonate (NaHCO3) that precipitates out in the first reaction is filtered out of the ammonium chloride (NH4Cl) solution, and the solution is then reacted with the quicklime (calcium oxide (CaO) left over from heating the limestone in step two. Step 3: 2NH4Cl + CaO → 2NH3 + CaCl2 + H2O CaO makes a strong basic solution. The ammonia from step three is recycled back to the initial brine solution of step one. The sodium bicarbonate (NaHCO3) precipitate from step one is then converted to the final product, sodium carbonate (Na2CO3), by calcination at 160-230 degrees celcius, producing water and carbon dioxide as byproducts: Final Step: 2NaHCO3 → Na2CO3 + H2O + CO2 The carbon dioxide from step four is recovered for re-use in step one. |
|