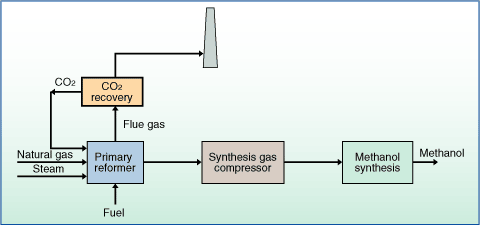
What is The methanol production Process?Methanol is a very useful form of alcohol, commonly found in fuels, as well as formaldehyde. The synthesis of methanol is carried out by reacting hydrogen gas with carbon monoxide gas. The hydrogen and carbon monoxide gases are obtained through the oxidation of methane. In this reaction, methane is reacted with oxygen at a relatively low temperature(1 ATM), causing the reaction to favour this side with more moles, in this case the products. This reaction is effective for obtaining carbon monoxide and hydrogen gas because is delivers the 1:2 ration of carbon monoxide to hydrogen gas that is required for the synthesis of methanol. This reaction is represented by the equation below:
CH4(g) + 1/2O2(g) → CO(g) + 2H2(g) Once the carbon monoxide and hydrogen gas are obtained from the oxidation of methane, they are reacted together to form methanol gas. The reaction is represented by the equation below: CO(g) + 2H2(g) → CH3OH(g) This reaction is carried out at temperatures exceeding 250 degrees and at pressures between 5 and 10 MPa. This is done to create the maximum yield of products from the reaction, with the least amount of leftover reactants. By having this reaction occur at such high temperatures and pressures it causes the reaction to favour the side of the reaction with the fewer number of moles, which is the products in this case. |